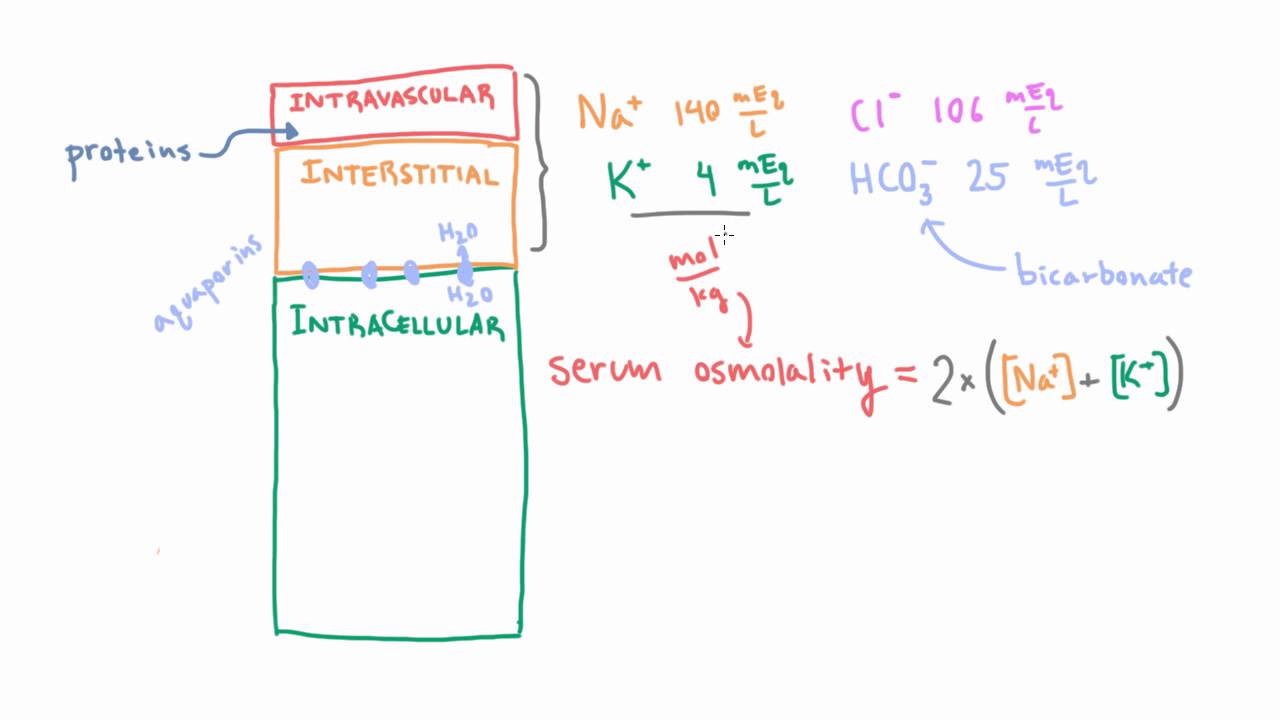
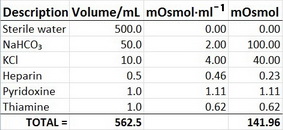
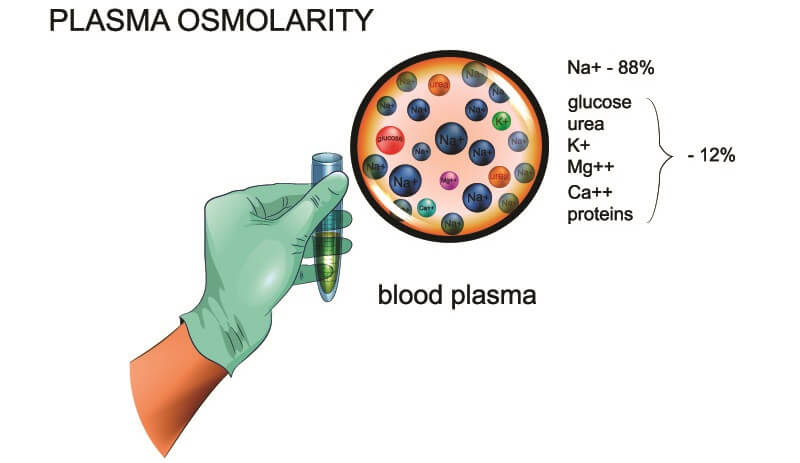
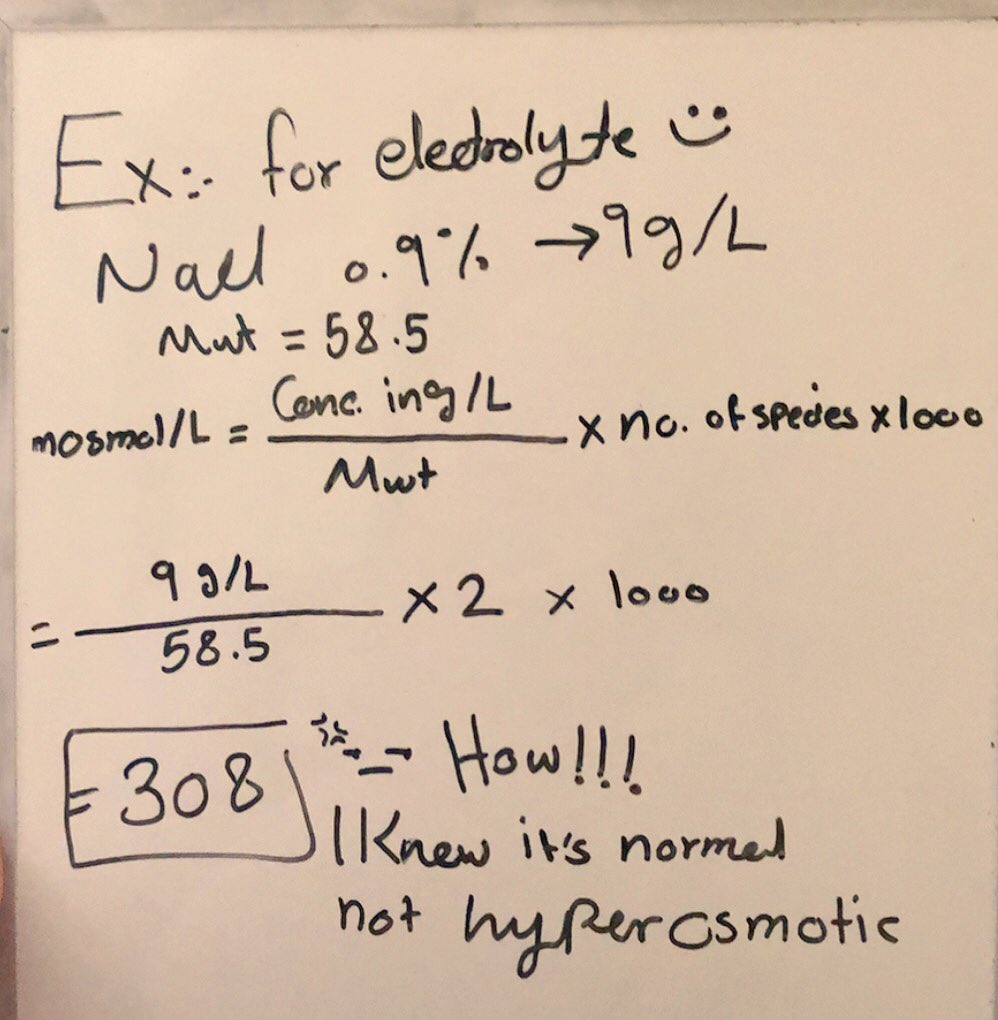Calculate your own osmolarity | Lab values and concentrations | Health & Medicine | Khan Academy - YouTube

PDF) Unmeasurable severe hypernatremia: A different way of using the calculated serum osmolality formula

Do you need help in solving osmolarity calculations? This video helps pharmacy students solve an osmolarity calculations question they are likely to... | By Pharmaceutical Calculations | Facebook
![SOLVED: Example Calculation of Plasma Osmolality Estimate asmolality from the following data: sodlium, 135 mEq/1 ; blood Mrd tbe plasma nlian 14 mg/dL; and glucose, 90 mg/dL [14 mgAL] [90 mgldL] osmolality SOLVED: Example Calculation of Plasma Osmolality Estimate asmolality from the following data: sodlium, 135 mEq/1 ; blood Mrd tbe plasma nlian 14 mg/dL; and glucose, 90 mg/dL [14 mgAL] [90 mgldL] osmolality](https://cdn.numerade.com/ask_images/ec9448d820484bc1bd864230d5283d50.jpg)
SOLVED: Example Calculation of Plasma Osmolality Estimate asmolality from the following data: sodlium, 135 mEq/1 ; blood Mrd tbe plasma nlian 14 mg/dL; and glucose, 90 mg/dL [14 mgAL] [90 mgldL] osmolality