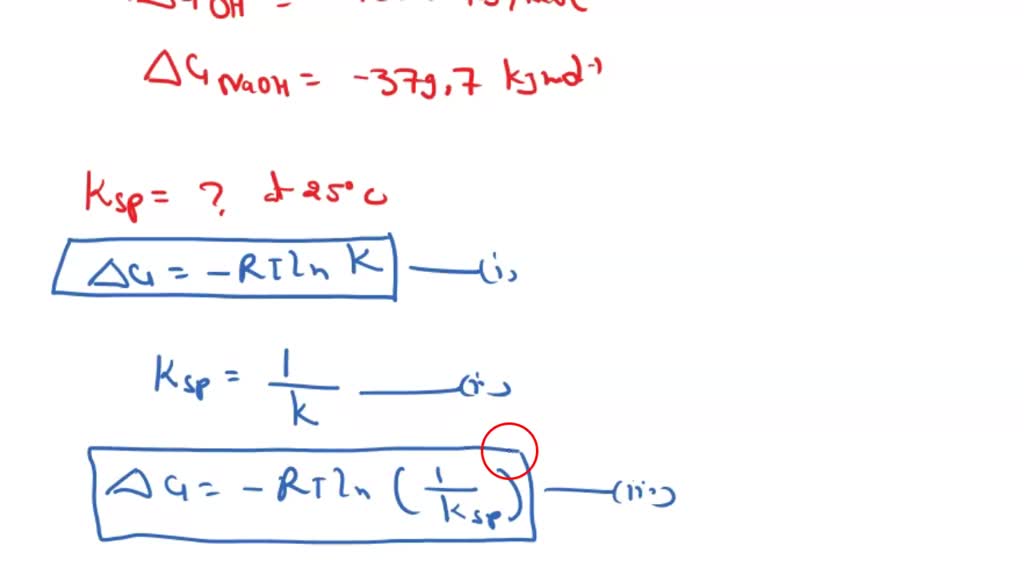
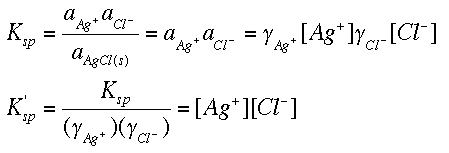SOLVED: The solubility of calcium sulfate (CaSO4) is 0.67 g/L. Calculate the value of Kps for calcium sulfate. If we know the solubility of CaSO4 and are asked to calculate his Kps.
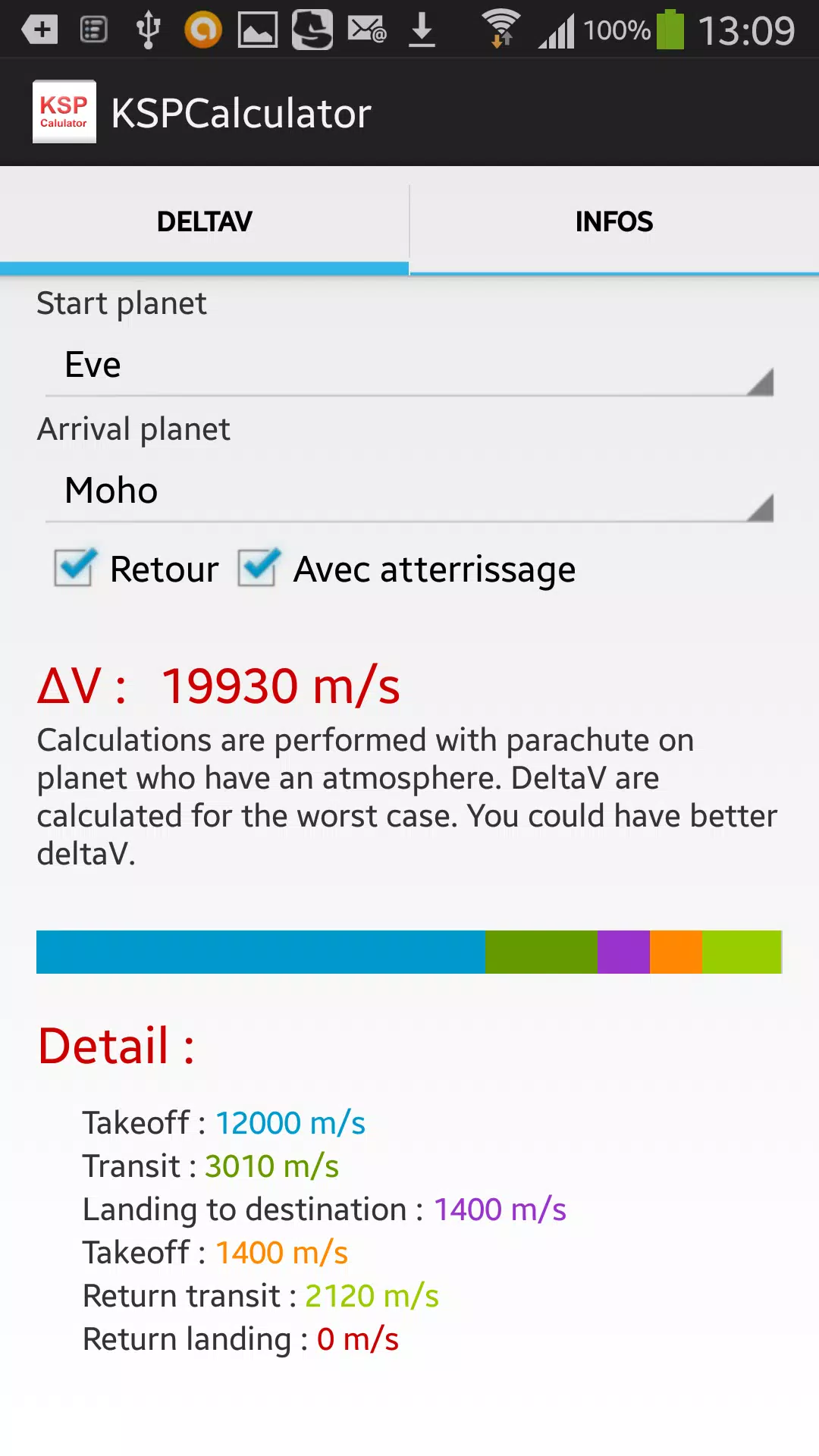
GitHub - Kerbas-ad-astra/ksp-calculator: Automatically exported from code.google.com/p/ksp-calculator
GitHub - Blaarkies/ksp-visual-calculator: Online tool for Kerbal Space Program that helps players determine delta-v requirements for a mission, and what type of comms dishes a certain satellite requires.

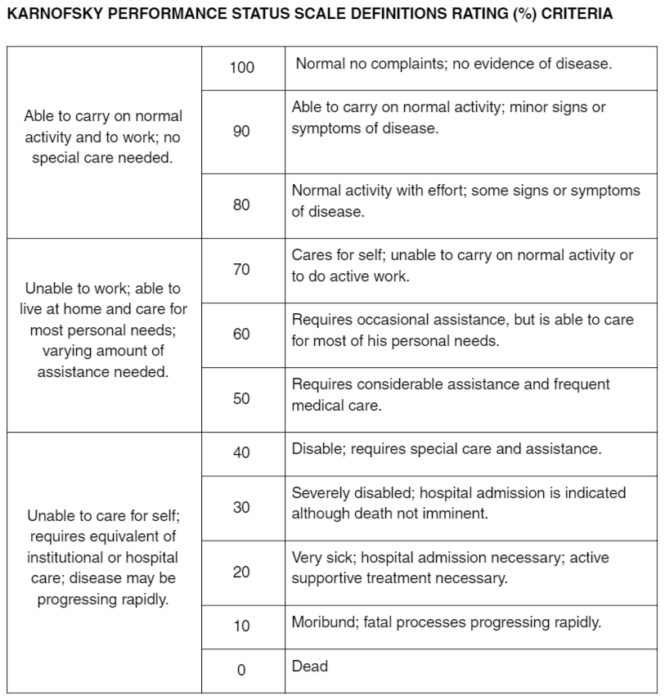
Karnofsky Performance Status (KPS) and Eastern Cooperative Oncology... | Download Scientific Diagram