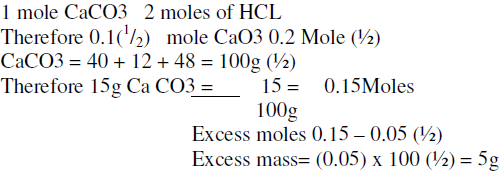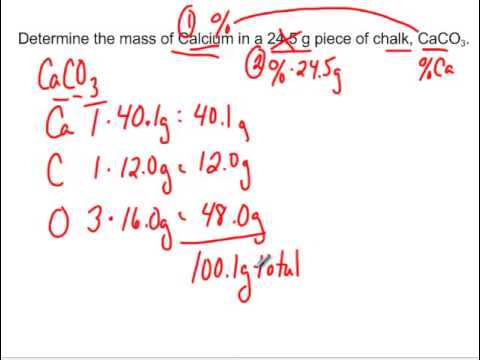Calculate the mass per cent of each element present in the molecule of calcium carbonate - CBSE Class 9 Science - Learn CBSE Forum

Question Video: Calculating the Mass of Calcium Carbonate Required to Produce a Given Mass of Calcium Oxide | Nagwa
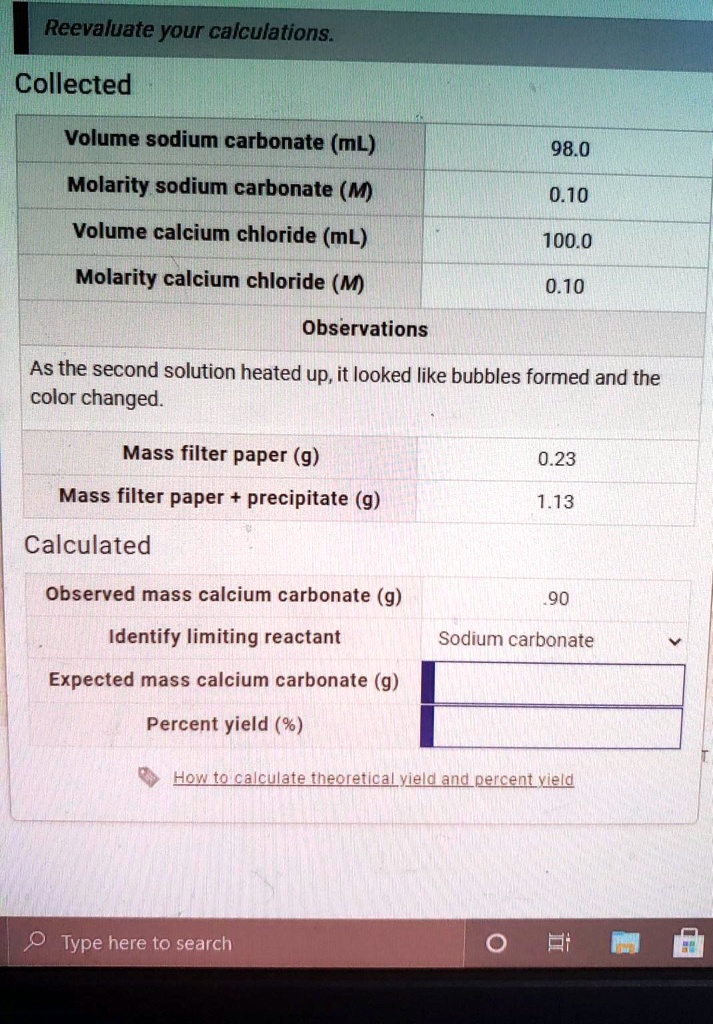
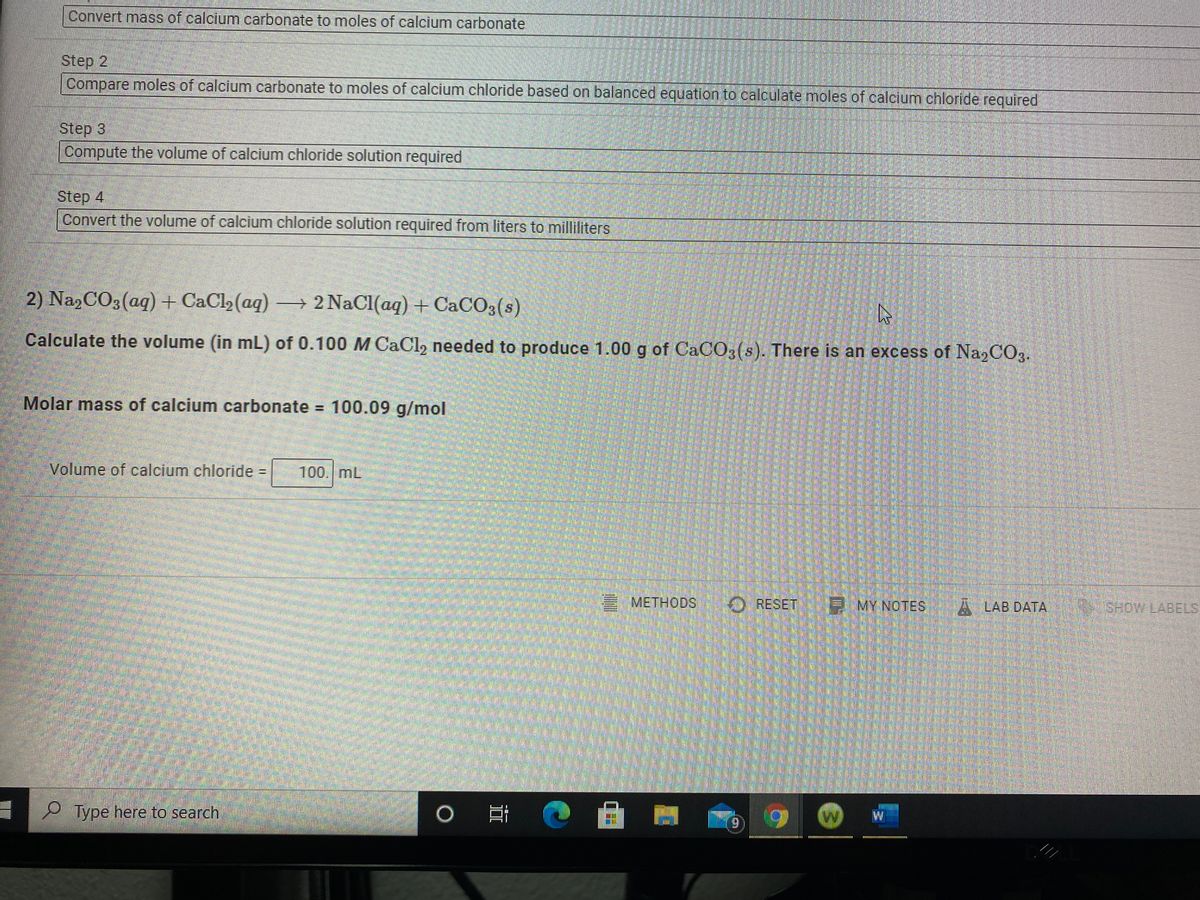
SOLVED: Reevaluate your calculations: Collected Volume sodium carbonate (ML) 98.0 Molarity sodium carbonate (M 0.10 Volume calcium chloride (mL) 100.0 Molarity calcium chloride (M) 0.10 Observations As the second solution heated up,it
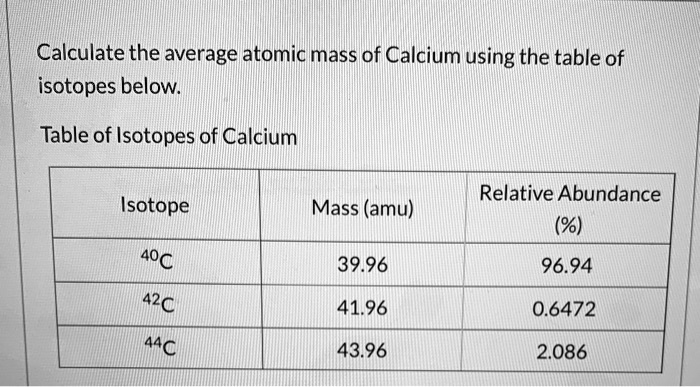
SOLVED: Calculate the average atomic mass of Calcium using the table of isotopes below: Table of Isotopes of Calcium Relative Abundance (%) Isotope Mass (amu) 40C 42C 39.96 96.94 41.96 0.6472 44C 43.96 2.086

Calculate the mole ratio of `240 g` of calcium and `240 g` of magnesium. `(Ca = 40 , Mg = 24 u)` - YouTube

equilibrium - How can I analytically calculate the rate of thermal decomposition of calcium carbonate? - Chemistry Stack Exchange