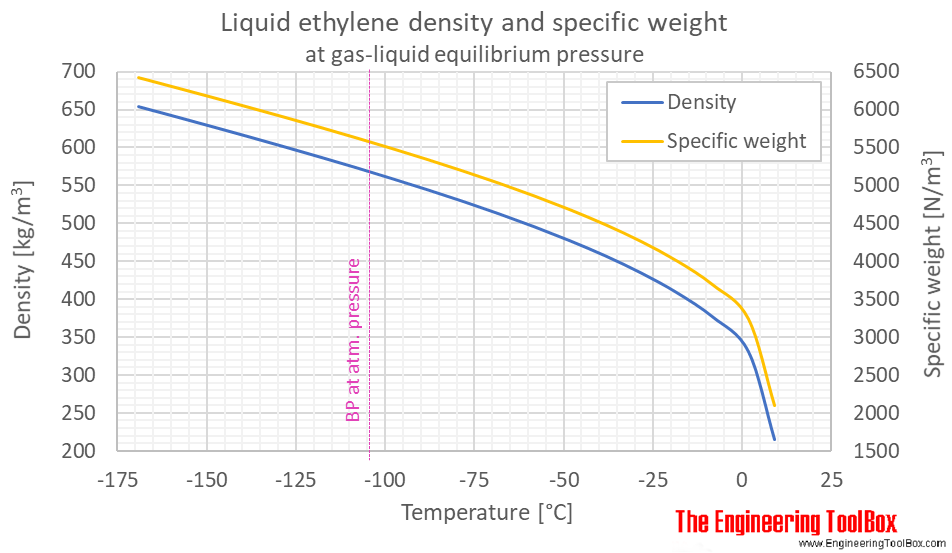
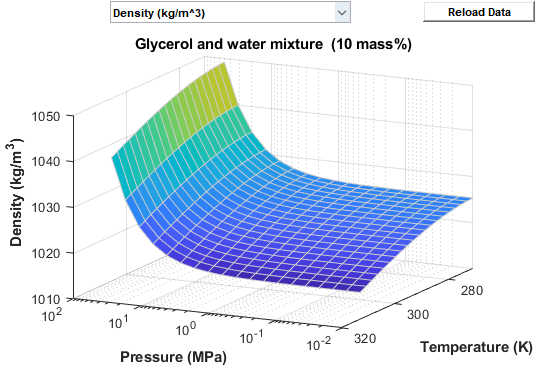
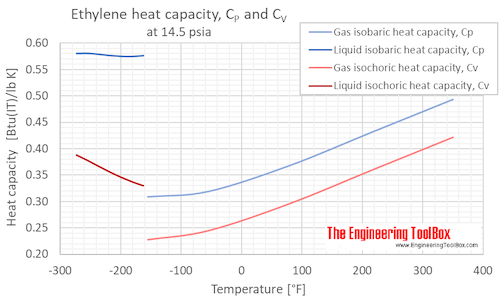
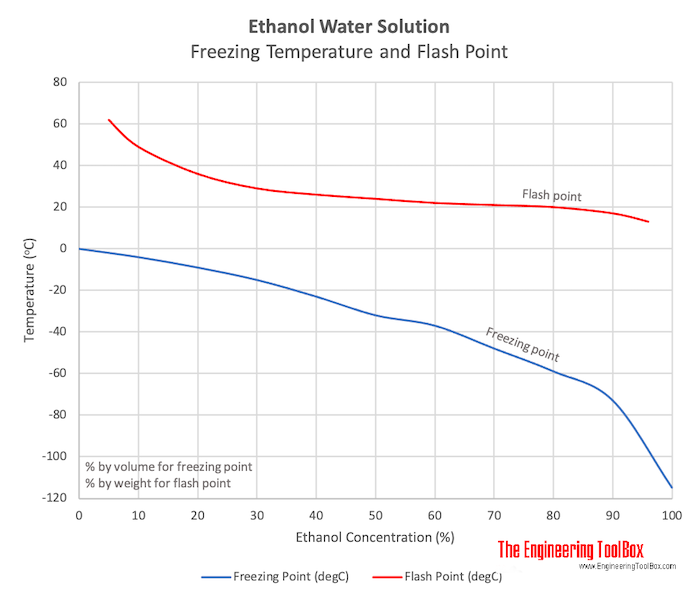
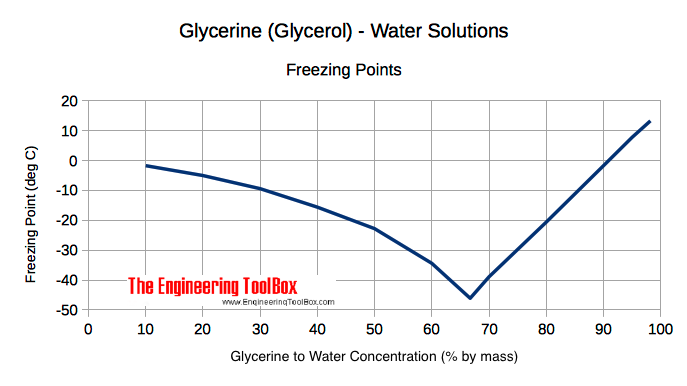
![PDF] Ethylene Glycol and Its Mixtures with Water and Electrolytes: Thermodynamic and Transport Properties | Semantic Scholar PDF] Ethylene Glycol and Its Mixtures with Water and Electrolytes: Thermodynamic and Transport Properties | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/b802f6702e8678ea6b78b956f251743775579bf2/3-Table1-1.png)
PDF] Ethylene Glycol and Its Mixtures with Water and Electrolytes: Thermodynamic and Transport Properties | Semantic Scholar

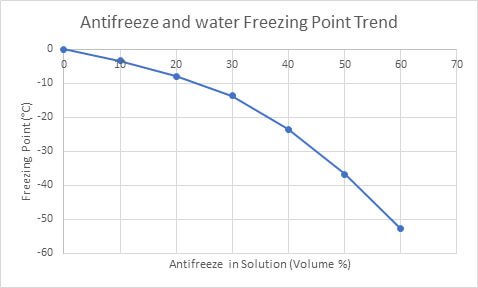
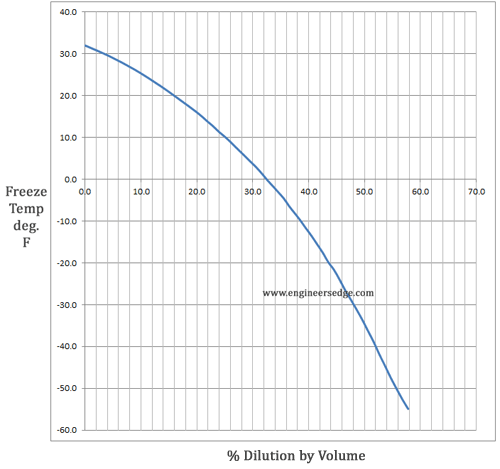
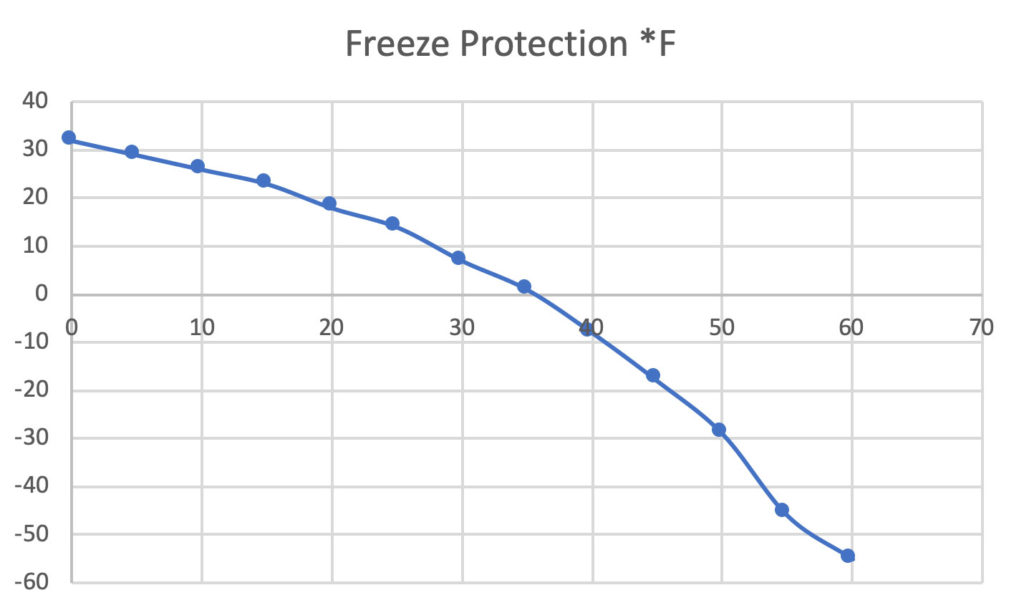
An antifreeze solution is prepared from 222.6 g of ethylene glycol (C2H6O2) and 200 g of water. Calculate the molality of the solution. If the density of the solution is 1.072 g

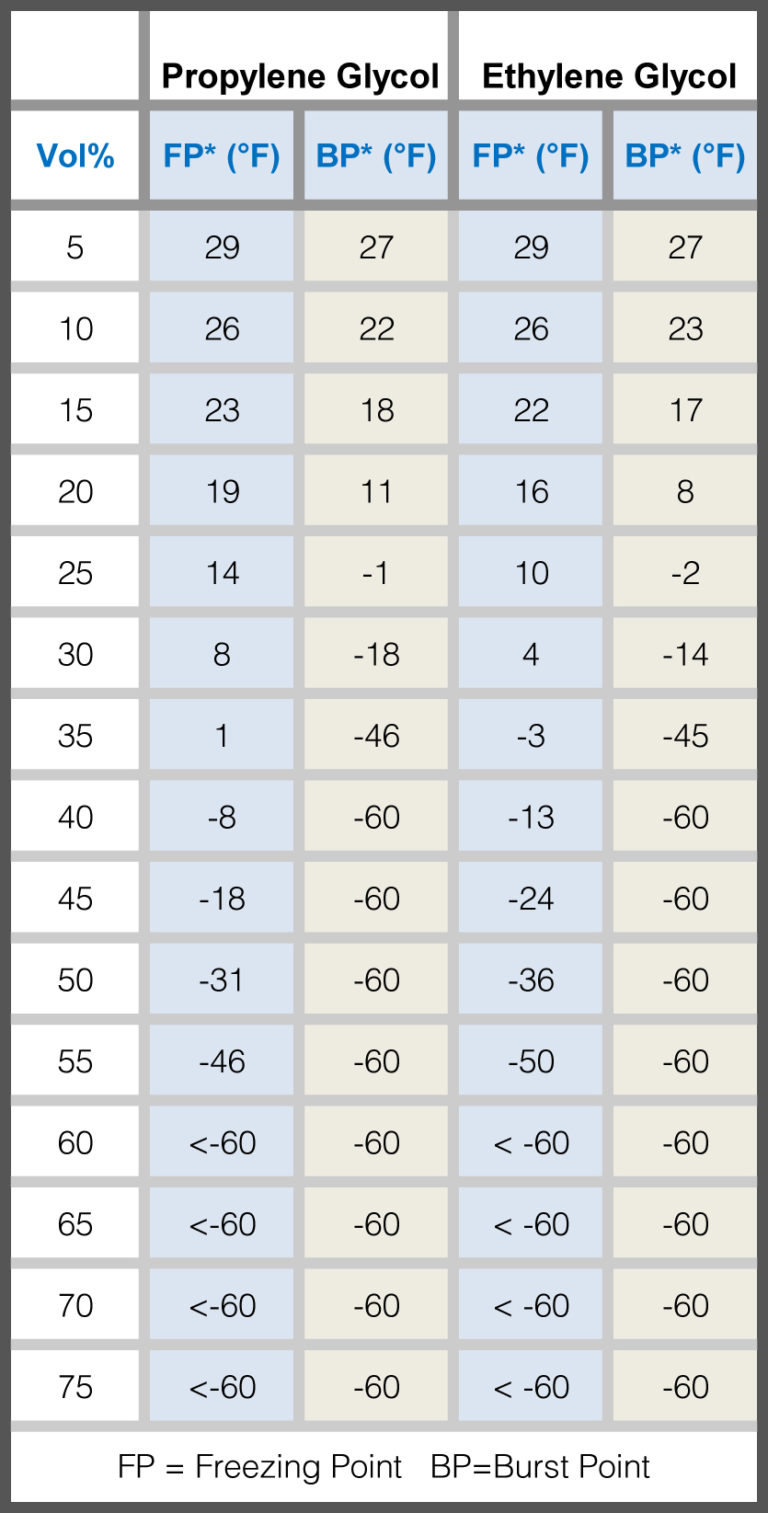
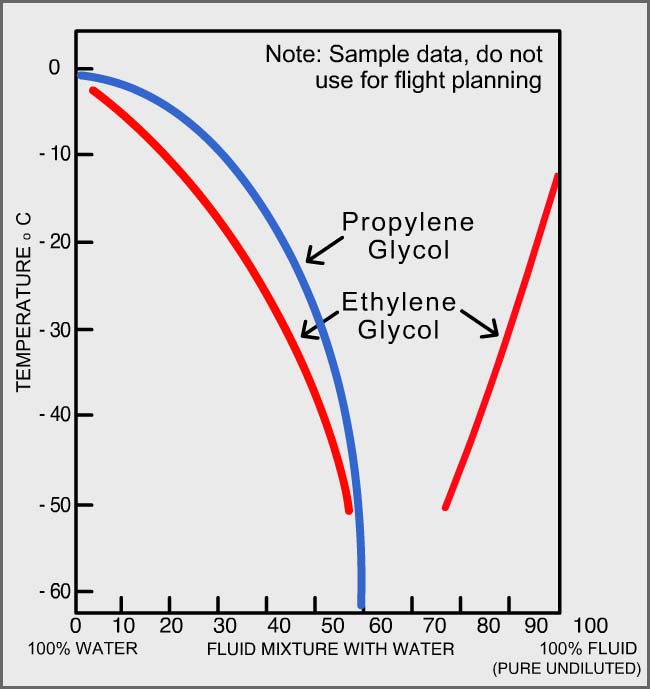
Physical properties of of the coolant liquid (55/45 by mass ethylene... | Download Scientific Diagram
![PDF] Ethylene Glycol and Its Mixtures with Water and Electrolytes: Thermodynamic and Transport Properties | Semantic Scholar PDF] Ethylene Glycol and Its Mixtures with Water and Electrolytes: Thermodynamic and Transport Properties | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/b802f6702e8678ea6b78b956f251743775579bf2/5-Table2-1.png)